|
公司基本資料信息
|
|||||||||||||||||||||||||||||
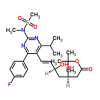
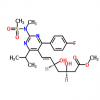
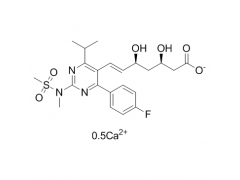
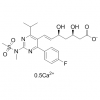
Synonym: 6-Heptenoic acid, 7-[4-(4-fluorophenyl)-6-(1-methylethyl)-2-[methyl(methylsulfonyl)amino]-5-pyrimidinyl]-3,5-dihydroxy-, calcium salt (2:1), (3R,5S,6E)-;(3R,5S,6E)-7-[4-(4-Fluorophenyl)-6-isopropyl-2-[methyl(methylsulfonyl)amino]pyrimidin-5-yl])-3,5-dihydroxyhept-6-enoic acid hemicalcium salt;Rosuvastatin Calcium(W.S);ZD 4522 Calcium;ROSUVASTATIN CALCIUM (ROSUVASTATIN HEMICALCIUM);Rosuvastadine;Rosuvastatin CalciuM (3R,5S,6E)-7-[4-(4-fluorophenyl)-6-(1-Methylethyl)-2-[N-Methyl(n-Methylsulfonyl)aMino]-5-pyriMidinyl]-3,5-dihydroxy-6-Heptenoic calciuM;Rosuvastatin Calium
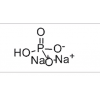
MF: C44H54CaF2N6O12S2
MW: 1001.14
Product Category: Inhibitors;Active Pharmaceutical Ingredients;Aromatics;Chiral Reagents;Sulfur & Selenium Compounds;HMG-COA reductase, Hypolipidemic drugs;rosuvastatin calcium R;API;Intermediates & Fine Chemicals;Pharmaceuticals;Statins’Anti-Lipidemia Agents;Rosuvastatin;DERMATOP
Melting Point: 122°C
Alpha: D24 +14.8° (c = 1.012 in 50% methanol)
Storage Temp.: -20°C Freezer
Form: powder
Color: white to beige
Optical Activity: [α]/D +12 to +18°, c = 1 in methanol: water (1:1)
λmax: 243nm(Phosphate buffer sol.)(lit.)
Merck: 14,8270
RTECS: MJ9675070
Anti-hyperlipidemia Drugs; Rosuvastatin calcium is an anti-hyperlipidemia drug which belongs to the inhibitor of HMG-CoA reductase successfully developed by the British AstraZeneca company. It is suitable for the treatment of various lipid abnormalities, including hypercholesterolemia, mixed lipid qualitative abnormalities and simple hypertriglyceridemia. Rosuvastatin calcium is listed as the strongest and the most comprehensive statins drugs which has entered into market for lipid-lowering and adjustment. Compared with atorvastatin which is currently recognized with the world's best efficacy, it has a better effect of lowering the level of LDL cholesterol and increasing the level of high-density lipoprotein. Moreover, it has a better tolerance, lower side effects and unique pharmacokinetic characteristics with the half-life about 20h. It only needs to be taken once a day.
Pharmaceutical Effects: Rosuvastatin is a selective inhibitor of HMG-CoA reductase. HMG-CoA reductase inhibitor is the rate-limiting enzyme of the transition hydroxy-3-methylglutaryl coenzyme A to valerate A (The precursor of cholesterol). The main site of action of rosuvastatin is liver-the target organs of cholesterol-lowering. Rosuvastatin increases the number of hepatic LDL receptors on the cell surface, promoting the absorption and catabolism of LDL, inhibits hepatic synthesis of VLDL, thereby reducing the total number of VLDL and LDL particles.
For patients of homozygous and heterozygous familial hypercholesterolemia, non-familial hypercholesterolemia, and mixed dyslipidemia, Rosuvastatin can lower the total cholesterol, LDL-C levels, ApoB levels, non-HDL-C levels. Rosuvastatin also reduces the level of TG and increase the level of HDL-C. For patients of pure hypertriglyceridemia, rosuvastatin can lower total cholesterol, LDL-C, VLDL-C, ApoB, non-HDL-C, TG levels and increase the HDL-C levels.
based on the general safety of pharmacology, repeated dose toxicity, potential genetic toxicity and the clinical information of carcinogenicity, no special toxicity was found for rosuvastatin on the human body. For studies on rat before and after birth, Rosuvastatin has obvious reproductive toxicity. It reduces the size, weight of the litter and birth rate. Treating female rats with a toxic dose which causes several times higher systemic exposure levels than the therapeutic exposure can enable the observation of these phenomena.
Pharmacokinetics: The drug can be absorbed in great amount by liver after oral administration with the volume of distribution at 134 L. After 3~5 hours, plasma concentration reaches the peak. The absolute bioavailability is around 20%. Plasma protein binding rate (mainly albumin) is about 90%. about 90% of the total dose of rosuvastatin is excreted by feces as prototype (including the absorption of the active substance and unabsorbed), with the rest excreted through the urine. 5% rosuvastatin in urine is in the form of prototype. Half-life of plasma elimination is about 19 hours. Elimination half-life doesn’t increase with increased dose. The geometric mean of plasma clearance rate is about 50L/hour (coefficient of variation 21.7%). Same as other HMG-CoA reductase inhibitors, the uptake of rosuvastatin by liver involves the participation of membrane transporter OATP-C. The transporter is important in the liver for the clearance of rosuvastatin.
Drug Interactions: 1. Cyclosporine: when this product is used in combination with cyclosporine, the AUC of rosuvastatin is averagely 7 times higher than that observed in healthy volunteers (compared with the same dose using this service). Combination usage does not affect the plasma concentrations of cyclosporine.
2. Vitamin K antagonists: same as other HMG-CoA reductase inhibitor, for patients of simultaneously usage of vitamin K antagonists (such as warfarin), at the beginning or gradually increasing the dose of this product may cause the increase of INR (international normalized ratio). Stop using the product or gradually decreasing the dose can lead to decrease in INR. In this case, appropriate INR testing is needed.
3. Combination of gemfibrozil, fenofibrate, other fibrates and lipid-lowering doses (≥1g/day) of niacin with HMG-CoA reductase inhibitors can increase the risk of myopathy, which may be due to that they can cause myopathy when given alone.
4. Antacids: Mixing the product and one antacid containing aluminum hydroxide and magnesium for suspension can reduce the concentration of statin rosuvastatin plasma by 50%. Taking antacids two hours after taking this can further reduce the impact. The clinical significance of this drug interaction has not been studied.
5. Erythromycin: Combination with erythromycin can cause rosuvastatin AUC (0~t) decrease by 20% and Cmax decrease by 30%. This interaction may be due to the gastrointestinal motility caused by increase of erythromycin.
6. Oral administration of contraceptives/hormone replacement therapy (HRT): while using this drug together with oral contraceptives can cause the increase of ethinyl estradiol and norgestrel AUC by 26% and 34%, respectively. When choosing the dose of oral contraceptives, the patients should consider the increase of the plasma concentration of these drugs. Yet there are no pharmacokinetic data on taking rosuvastatin and HRT together. Therefore, we cannot exclude the presence of similar interactions. However, in clinical trials, this kind of combination is widely applied and can be well tolerated by patients.
Safety: In terms of safety issue, the incidence of adverse cases for rosuvastatin calcium was similar to that of other related drugs. Adverse cases include elevated ALT, increased GGT and nausea.
Side Effects: 1. Adverse reactions of this product are generally mild and transient such as common headache, dizziness, constipation, nausea, abdominal pain, muscle pain and weakness.
2. For observed patients, the increase of creatine kinase (CK) levels is dose-related; the majority of cases were mild, asymptomatic and transient. Stop the treatment once the CK levels elevate (> 5 × ULN).
3. Impact on the liver: for a small number of patients, it was observed for a drug dose-related increase in transaminases; the majority of cases were mild, asymptomatic and transient.
Uses: For making lipid-lowering drugs.
Description: Rosuvastatin, one of the two new statins launched for the treatment of hypercholesterolemia, has high hepato-selectivity and more potent inhibitory effect on HMG-CoA reductase than the previously marketed statins. In rat hepatocytes, it inhibits cholesterol biosynthesis with an IC50 of 1.12 nM, which is ~100-fold higher potency than pravastatin. Rosuvastatin is synthesized in a 12-step sequence, entailing the construction of a pyrimidinyl aldehyde intermediate in eight steps and subsequent introduction of the dihydroxyheptenoate side chain via Wittig reaction with a bketophosphorane reagent and stereoselective carbonyl reduction of the resultant enone. Pharmacokinetic properties of rosuvastatin in humans, dosed at 5–80 mg, are approximately linear with dose. Following oral administration, rosuvastatin is rapidly absorbed with an oral bioavailability of ~20% and tmax of ~3 h. It has a prolonged duration of action, with terminal t1/2 of ~20 h, compatible with once-daily dosing. In humans, rosuvastatin is minimally metabolized through CYP2C9 and CYP2C19, with little or no metabolism via the CYP3A4. Approximately 90% of the administered oral dose is eliminated in the feces (92% as the parent compound) and the rest in the urine. Rosuvastatin is considered a “superstatin” due to its ability, at well-tolerated doses, to lower LDL cholesterol and triglycerides to a much greater extent than first generation statins. In patients with hypercholesterolemia, rosuvastatin treatment at doses of 5 and 10 mg/day over 12-week period resulted in 40–43% reduction of LDL-cholesterol levels, 12–13% increase in HDL-cholesterol, and 17– 19% reduction in triglycerides. In comparison, the efficacy range of LDL-cholesterol reductions by atorvastatin (10 mg/day), pravastatin (20 mg/day), and simvastatin (20 mg/day) was 28–35%. Rosuvastatin is a well-tolerated drug at doses of 1–20 mg and the most common side effects at these doses are headache, myalgia, pain and pharyngitis, which are consistent with those previously reported for statin therapy.
Chemical Property: White to Off-White Crystalline Solid
Uses: A selective, competitive HMG-CoA reductase inhibitor. Antilipemic.
Uses: antiinflammatory, glucocorticoid
Uses: antirheumatic
Definition: ChEBI: An organic calcium salt that is the hemicalcium salt of rosuvastatin.



 通過認證
通過認證